Reprogramming Veterans' Blood Cells to Study Gulf War Illness

More than a quarter of the 700,000 veterans who served in the 1991 Gulf War are still suffering from chronic fatigue, memory loss, joint pain, insomnia and stomach problems. This cluster of unexplained symptoms is characteristic of Gulf War illness, now an official diagnosis recognized by the Veterans Administration, but one that still vexes doctors and researchers alike.
Now, with funding from the U.S. Department of Defense, scientists from Drexel University College of Medicine and Boston University are applying the most cutting-edge stem cell technology to solve this 25-year-old medical mystery.
Using blood cells obtained from 300 veterans in the Gulf War Illness Consortium [sites.bu.edu/gwic], Drexel College of Medicine researchers have genetically modified the cells into human induced pluripotent stem cell (hiPSC) lines. This means the cells behave like embryonic stem cells; once treated with various growth factors, they have the ability to form practically any other type of adult cell, from a kidney cell to a neuron.
Since these pluripotent cell lines continue to divide, the researchers were able to create a stem cell biorepository, which can be made available to any interested researcher to study the mechanisms of Gulf War illness and to test potential treatments. The cell lines will be especially groundbreaking for studying Gulf War illness, because they preserve the genetic and possibly epigenetic factors specific to disease susceptibility.
This paradigm shift in the study of Gulf War illness was the subject of a paper published by the researchers in the journal Neurology. 1
"The GWI research community does not have very good model systems in which to test hypotheses and potential therapeutics. These cell lines represent an important shift toward an experimental model that will be much more useful for understanding this disease," says Peter Baas, PhD, a professor in the Department of Neurobiology & Anatomy at Drexel University College of Medicine. "We see this as an urgent situation. These veterans are not getting any younger."
Baas and his team will be the first group in the world to use hiPSC lines derived directly from Gulf War veterans, comparing those who did or did not get sick after serving.
The cell lines will be used to identify how alterations in axonal transport, microtubule functioning and neuroinflammation may contribute to GWI symptoms. For instance, Baas's research group found that microtubules — cylinder structures that give shape to cells and power their movements — could be a prime target for treating this disease in a study published in the journal Traffic. 2 "In addition to being an architectural element that helps to shape the cell, the microtubule also acts as a railway, which transports organelles throughout the cytoplasm," says Baas. "We hypothesized that toxins would change the typical way microtubules are chemically modified in neurons, and that a drug like tubacin could restore the modifications to normal, thereby treating the disease."
The study's results suggest that dopamine alterations after toxin exposure are in part due to changes in microtubules, and restoring microtubule function to a more normal state could help to alleviate symptoms.
Baas hopes that the GWI stem cell biorepository will help his team refine their results and better understand this connection. He says that studying the illness could also provide insight into other neurodegenerative diseases. The use of organophosphate pesticides is widespread around the world, and growing evidence indicates a link between the pesticides and disorders such as Parkinson's disease.
"We're living in an increasingly toxic world," says Baas. "It's likely that this kind of disease is going to repeat itself if we don't educate ourselves as to its causes, as well as how to prevent and treat it."
DEVELOPING A CRITICAL TOOL
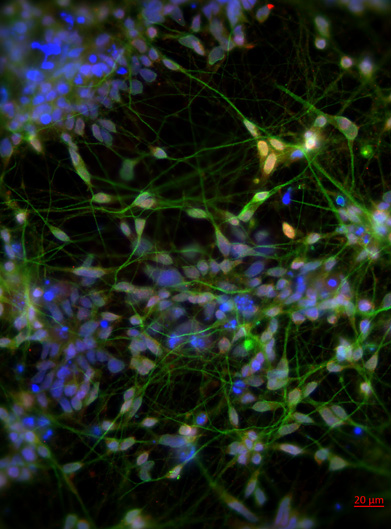
Human induced pluripotent stem cells (hiPSCs) were differentiated into mature neurons in order to study how Gulf War neurotoxicants alter microtubule behaviors in human neurons. In the magnified image used here as background, blue is DAPI, a nuclear stain; green is MAP2, a neuron specific marker enriched in dendrites and the cell body; and red is TBR1, a forebrain cortical neuronal specific transcription factor. After validation, the neurons can be used to model Gulf War illness in the culture dish.
After earning his PhD at Drexel University College of Medicine, Liang Qiang spent four years at Columbia University, converting adult tissue cells into neurons to study debilitating diseases like Alzheimer's and Parkinson's.
The innovative research caught the attention of Peter Baas, PhD, a professor in the Department of Neurobiology & Anatomy, who believed Qiang's expertise could be beneficial for investigating other neurological conditions.
Qiang has returned to his alma mater, now a research assistant professor using cutting-edge research to study Gulf War illness. Studying patient-derived cells offers a number of major advantages to understanding the origins of this disease, according to Qiang.
Japanese researcher Shinya Yamanaka, who won the Nobel Prize for his work in 2012, pioneered the extraordinary process of reprogramming adult cells to an embryonic-like state.
Although hiPSC lines were originally intended for clinical application, using them to cure sick patients has proved challenging. However, these cells have become a critical tool for investigating human disease and testing new treatments. In diseases ranging from Parkinson's to amyotrophic lateral sclerosis, hiPSC lines offer an invaluable method for studying illness, without the ethical concerns of cells derived from embryos.
Since hiPSC are human cells, they have human proteins and pathways that may not be reflected in animal models. And most importantly, Qiang says, these cells are derived from the patients themselves, which means they harbor the wide array of genetic factors that may contribute to the disease.
"The big advantage of using patient-derived cells is that many diseases cannot be traced to just one gene being mutated, but rather are due to the complex interactions of genes. Susceptibility to these diseases may also be based on epigenetic factors, which cannot be looked at in animal models," Qiang says. "These cell lines preserve the complete genetic composition of those affected by the disease."
Because pluripotent cell lines are essentially immortal, they can be used for study an unlimited number of times, including for comparison against results of parallel studies on other neurodegenerative diseases.
Notes:
1. Liang Qiang, MD, PhD; Anand N. Rao, PhD; Peter W. Baas, PhD; and colleagues at Boston University published "Reprogramming Cells From Gulf War Veterans Into Neurons to Study Gulf War Illness" in Neurology, May 16, 2017.
2. Anand Rao, PhD, and Ankita Patil (co-first authors); Zachary Brodnik, doctoral student; Liang Qiang, PhD; Rodrigo España, PhD; Peter Baas, PhD; and colleagues published "Pharmacologically Increasing Microtubule Acetylation Corrects Stress-exacerbated Effects of Organophosphates on Neurons" in Traffic, July 2017.
Back to Top