Research
Melanoma is the deadliest form of skin cancer, as patients presenting with metastatic disease have a five-year survival of ~25%. However, early clinical intervention prior to metastasis yields ten-year survival rates of ~92%. Our lab's overarching goals are to investigate mechanisms driving the metastatic spread of melanoma, elucidate the role of the tumor microenvironment in response to therapy, and uncover new therapeutic targets.
CADM1 biology
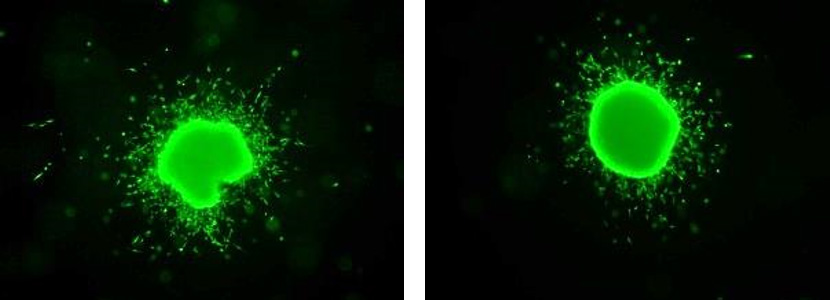
Melanoma spheroid invading into collagen.
Through transcriptomic analysis, we have identified cell adhesion molecule 1 (CADM1) as a suppressor of metastatic traits in human melanoma. CADM1 levels are inversely associated with tumor stage and high CADM1 levels correlate with better prognosis. We found that CADM1 inhibits melanoma disease progression by suppressing invasiveness and potently inducing non-adherent cell death — two properties critical to metastatic dissemination. Our group currently uses multi-disciplinary approaches to investigate the mechanisms by which CADM1 is regulated and how it signals. We are also interested in assaying the role CADM1 plays in melanoma immune surveillance and determining if CADM1 expression could be considered a diagnostic marker for immunotherapy efficacy.

CADM1 expressing melanoma cells.
Post-translational modifications and the tumor microenvironment
Metastatic dissemination relies on melanoma’s ability to escape immune surveillance and migrate to distant organs. These processes are heavily influenced by cell surface proteins, which can be regulated by post-translational modifications like protein glycosylation. One potential modified cell surface protein mediating these effects is cell adhesion molecule 1 (CADM1). We are examining how post-translational modifications on CADM1 and other proteins impact the tumor immune micro environment, which may untangle the complex relationship between glycosylation and melanoma immune evasion and metastasis.
Novel Therapeutics and Overcoming Resistance Mechanisms
A secondary focus of the laboratory is defining novel therapeutics, resistance mechanisms, and second-line therapy options for patients suffering from mutant BRAF-driven cancers. BRAF is a serine/threonine kinase in the RAS-RAF-MEK-ERK mitogenic pathway. Activating mutations in this pathway lead to many human malignancies. Notably, BRAF is altered in ~50% of melanomas, 60% of thyroid carcinomas, and ~10% of colorectal cancers. BRAF’s most prevalent mutation occurs at position 600, where valine is substituted for either glutamic acid or lysine (BRAFV600E/K). BRAFV600E/K mutant selective inhibitors have been developed and are currently in clinical use, but these drugs are only effective in certain settings, and patients who benefit from these agents often develop drug resistance leading to eventual relapse. We aim to study a "next generation" mutant BRAF inhibitor, PLX8394, currently in clinical trials. Our recent work suggests PLX8394 treatment uniquely breaks RAF level dimerization, which is important because existing FDA-approved RAF inhibitors are ineffective against mutant BRAF cancers with high RAF dimerization. Through in vitro, in vivo, and ex vivo experimentation, we will assay the effectiveness of PLX8394 in a panel of dimerization-dependent non-canonical mutant BRAF melanoma and mutant BRAF colorectal and thyroid cancers.

Overview of resistance mechanisms to RAF inhibitors in mutant BRAF melanoma.
Mutant BRAF melanoma spreads to the brain in nearly 50% of patients, which carries a poor prognosis and life expectancy of 4–6 months. Treatment options are limited by the unique challenges of the blood-brain barrier and the immune-privileged nature of the brain. We seek to better understand how treatments change the immune microenvironment of melanoma brain metastasis, with the goal of limiting minimal residual disease and improving durable response.
Vehicle
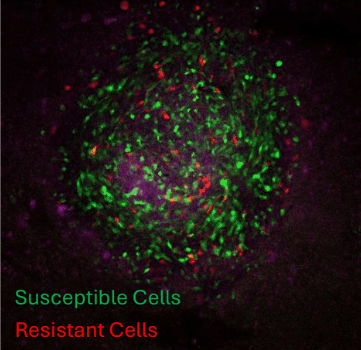
Dox
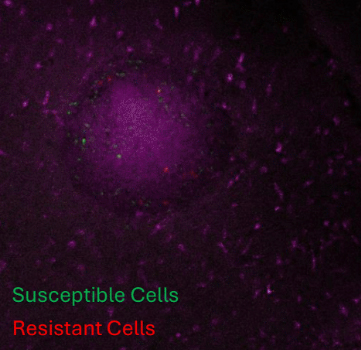
3D syngeneic melanoma spheroids on mouse forebrain slices. Green staining shows susceptible cells undergoing pyroptosis after treatment and red staining shows resistant cells.
D4M.3a

D4M.3a + pmel-1 T Cells

Syngeneic 3D model of mouse melanoma spheroids co-cultured with melanoma antigen-specific CD8+ T cells (pmel T cells). On the left, a spheroid alone without pmel, and on the right, spheroids incubated with pmel and imaged after 48 hours. Co-cultures were then imaged via confocal microscopy to determine infiltration efficiency.
Uveal melanoma
Uveal melanoma is the most common intra-ocular malignancy in adults with ~2000 cases per year in the United States and 5-6 million cases per year globally. About half of these patients will develop metastasis, primarily in the liver, and patients with hepatic metastasis generally have a one year survival outlook. There are few successful therapeutic options for liver metastatic uveal melanoma due to the unique environment of the liver, which is adept at clearing toxins. Uniquely, 80% of metastasizing uveal melanoma lesions have biallelic inactivation or deletion of BRCA1 associated protein-1 (BAP1). The lab is interested in the role BAP1 plays in metastasis and understanding how the liver microenvironment contributes to metastasis.

Intrasplenic injections of uveal melanoma in mice. Green signal shows GFP labeled uveal melanoma cells and red signal shows hepatic blood vessels.

Uveal melanoma spheroids on a mouse liver slice.
Back to Top