Largest Study of Malaria Genes Reveals a Finely Tuned Genome Full of Potential Drug Targets

- On the Trail of Deepfakes, Drexel Researchers Identify ‘Fingerprints’ of AI-Generated Video
- New AI-Technology Estimates Brain Age Using Low-Cost EEG Device
- Bolstered by Research Consortium with Drexel, Jefferson’s Sidney Kimmel Cancer Center Earns NCI Comprehensive Cancer Center Designation
- Alyssa Kemp Named as 2024 Truman Scholar
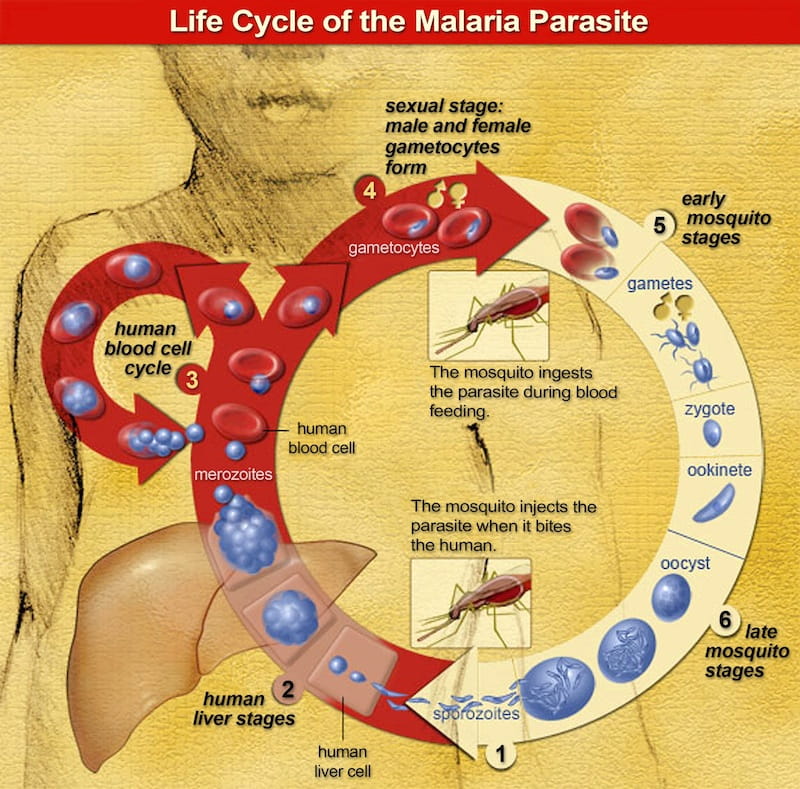
The blood stage parasites cause the malaria symptoms. When certain forms of blood stage parasites ("gametocytes") are picked up by a female Anopheles mosquito during a blood meal, they start another, different cycle of growth and multiplication in the mosquito. (NIAID)
The malaria parasite is a fine-tuned machine consisting primarily of essential genes, scientists at the Wellcome Trust Sanger Institute, Drexel University and their collaborators have found in the first ever large-scale study of its kind.
Scientists analyzed more than half of the parasite’s genes and discovered that 1,196, or two thirds of its genome, are essential for survival. This represents the largest proportion of essential genes found in any organism studied to date.
The results, published today in Cell, identify many potential targets for new antimalarial drug development. Nearly half of the world’s population is at risk of malaria and more than 200 million people are infected each year. Establishing new gene targets is essential for combatting the life-threatening disease, since malaria parasites are quickly developing widespread resistance to antimalarial drugs.
Plasmodium parasites, which cause malaria, are ancient organisms and around half their genes have no similar genes in any other organism, making it difficult for scientists to analyze their function.
To overcome this barrier, the research team used a cutting-edge method called DNA Barcoding, which involves tagging specific disrupted genes with molecular barcodes. These barcodes enable scientists to identify individual mutants and measure the growth of the genetically modified parasite by counting the number of barcodes using a benchtop DNA sequencer.
The researchers studied the genes in Plasmodium berghei, a species of malaria parasite that infects mice, and then “knocked out” or switched off 2,578 genes — more than half of its genome. The team then used next generation genome sequencing technology to count those barcodes, and measure the growth of each genetically modified malaria parasite inside mice.
If a gene was not essential, then the parasite grew, but if the knocked-out gene was essential, the parasite disappeared.
The results show that the malaria parasite can easily dispose of the genes that reveal its presence to the host’s immune system. This poses problems for the development of malaria vaccines as the parasite can quickly alter its appearance to the human immune system, and as a result the parasite can build resistance to the vaccine.
“Our study found that below the surface the parasite is more of a Formula 1 race car than a clunky people carrier. The parasite is fine-tuned and retains the absolute essential genes needed for growth,” said Julian Rayner co-lead author on the study and researcher at the Sanger Institute. “This is both good and bad. The bad news is it can easily get rid of the genes behind the targets we are trying to design vaccines for, but the flip side is there are many more essential gene targets for new drugs than we previously thought.”
Akhil Vaidya, PhD, a professor, and Michael Mather, PhD, an assistant professor at Drexel University College of Medicine, provided the Sanger Institute team with data analysis to better understand the genes that function in the parasite’s mitochondria and to identify “druggable” pathways within the parasite.
Vaidya’s group is one of the world’s leading centers for malarial research, which has been investigating mitochondrial functions in malaria parasites for over two decades.
“The identification of 1,196 genes likely to contribute to normal parasite growth provides us with crucial leads of the essential genes that we can investigate further,” Vaidya said. “We now have the opportunity to uncover the biological pathways crucial to this process, which will help validate targets for future drugs.”
In This Article
Contact
Drexel News is produced by
University Marketing and Communications.